FDA Set To Approve Pfizer Vaccine For Kids 12-15 As Early As Next Week
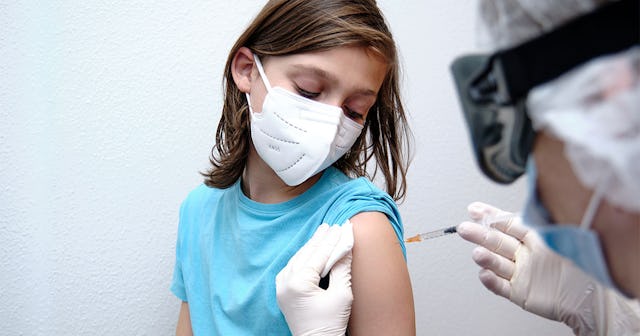
The FDA is expected to grant Pfizer emergency use authorization for its COVID vaccine to be given to kids 12 and up by next week
Daily vaccine rates in the U.S. have started to slow — a good indication that many of the adults who want to get vaccinated already have. All adults are eligible, and data shows that around half of adults have received at least one dose of a COVID shot, with nearly one third of the adult population being fully vaccinated. There’s been just one more major milestone to wait for: For a shot to be approved for kids, especially with the fall school year looming on the horizon.
Now, there’s good news. Pfizer, one of the first companies to receive emergency use authorization for its two-shot vaccine, has already applied for authorization to start giving the shot to adolescents, ages 12 and up. Now, experts say that authorization could come within the next week.
Pfizer announced at the end of March that it had submitted data from a clinical trial of nearly 2,300 adolescents between 12 and 15 years old. Half the kids received the two-dose regimen that has already been given to many adults, and half were controls who received placebo shots. Pfizer reported that the vaccine triggered a stronger immune response in teens than it does in young adults, and that there were only 18 cases of COVID-19 reported among trial participants—all of them in kids who received the placebo. The pharmaceutical company says that those results indicate the shot provides similar protection to kids as it does to the adults who have been receiving it for months.
This is great news. Even though children are statistically less likely to suffer from severe COVID-19, there have still been around 300 kids who have died from the virus in the U.S. Nearly 600,000 total people have died of COVID since the start of the pandemic.
If the FDA grants emergency use authorization to the Pfizer vaccine, the next step would be for a CDC panel to meet and create recommendations for how it should be distributed, and whether any kids, such as those with underlying health conditions, should be prioritized to receive the vaccine first.
Moderna, another company that created a vaccine that’s currently being used in adults, is currently conducting clinical trials of its vaccine in teens and younger kids. Results from those trials are expected to come sometime during the summer.