Meet The First Person To Get A Fully Approved COVID-19 Vaccine
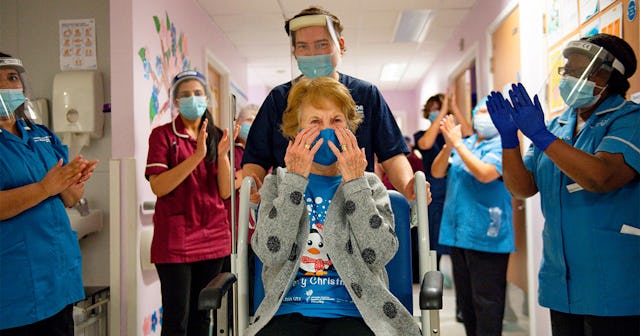
90-year-old Maggie Keenan became the first person in the world to receive a coronavirus vaccine that was approved in the West after clinical trials
Finally, there’s a light at the end of the tunnel. Multiple pharmaceutical companies have announced results from their clinical trials for newly developed COVID-19 vaccines, showing that their candidates are safe and even more effective than many scientists hoped. Now, the first vaccine, from Pfizer, has been approved by regulatory agencies for use on the general public, and the U.K. has begun its vaccination program to end the pandemic. Meet the first person in the western world to receive a shot against COVID: Maggie Keenan.
90-year-old Keenan (who will actually turn 91 next week) is a former jewelry shop assistant who retired just four years ago. She’s also a mother of two and a grandmother of four. She lives in Coventry, and has spent most of 2020 quarantining because her age puts her at such high risk for severe COVID-19.
JACOB KING/POOL/AFP via Getty Images)
She received her vaccine at University Hospital in Coventry at 6:31 a.m. local time, marking the first shot in a program to vaccinate as much of the population as possible in the U.K. COVID-19 has infected nearly 2 million people in the country, and more than 61,000 have died there.
“I feel so privileged to be the first,” Keenan told reporters after getting her shot, adding that being vaccinated means she can “finally look forward to spending time with my family and friends in the new year after being on my own for most of the year.”
JACOB KING/POOL/AFP via Getty Images
She also encouraged others to do the same thing she’s doing. “My advice to anyone offered the vaccine is to take it — if I can have it at 90, then you can have it, too,” she said.
As Keenan was rolled down a hospital hallway after receiving her shot, hospital staff members, in their PPE, gave her a round of applause.
The Pfizer vaccine that’s being distributed in the U.K. is expected to be granted emergency use authorization in the U.S. in a matter of days. Soon after that, the FDA is expected to grant emergency approval to another candidate, made by Moderna. Experts say healthcare workers in the U.S. could start receiving shots as soon as next week, and are hopeful that enough people will take it to restore life to some normalcy by the end of 2021.
There are a number of other promising vaccine candidates being developed all over the world, lending some hope that the COVID-19 pandemic might actually come to an end. That will depend on people getting their shots, though, and continuing to take other preventative measures, like masking and social distancing, in the meantime.